Abstract Introduction:
Maintaining safe working environments for health care personnel, especially for those who regularly handle hazardous drugs (HDs), is of utmost importance. Studies have shown that when closed system transfer devices. (CSTDs) are used with standard open barrel syringes, cyclophosphamide (CP), a commonly used HD, is transferred to the syringe plunger during compounding or administration processes. This contamination can then be transferred to the work environment, endangering workers.
Purpose:
The purpose of this study was to quantify HD contamination of the inner surface of standard open barrel syringes and to compare contamination levels between three commonly used HDs: 5-fluorouracil (5-FU), CP, and ifosfamide (IF).
Methods:
Each HD was transferred from a vial to an intravenous (IV) bag using a standard open barrel syringe and Becton, Dickinson and Company (BD) PhaSealTM CSTD connectors. Samples were taken from the inner surface of each of the syringe barrels to measure the amount of HD contamination. Each drug was tested 15 times and compared to a positive control.
Results:
Significant amounts of each drug were transferred to the inner surfaces of the syringes. The average amounts of each drug measured were: 5-FU, 1327.7 ng (standard deviation [SD] =873.6 ng); CP, 1074.8 ng (SD=481.6 ng); and IF, 1700.0 ng (SD =1098.1 ng). There was no statistically significant difference between the three drugs (p=0.14).
Conclusion:
This study underscores the presence of HD contamination on standard open barrel syringe inner surfaces after transfer of drug from vial to syringe to IV bag. Such contamination could be spread in the working environment and expose health care workers to harm.
1. Introduction
Hazardous drugs (HDs), as defined by the National Institute for Occupational Safety and Health (NIOSH), are Food and Drug Administration approved medications that meet certain toxicity criteria for humans or animals. These drugs have been determined to be carcinogenic, reproductively toxic, developmentally toxic, genotoxic, and/or toxic to specific organs (e.g., heart, lungs, kidneys, liver).1,2 Common HDs include antineoplastic agents (e.g., 5-fluorouracil [5-FU], cyclophosphamide [CP], and ifosfamide [IF]), nucleosides and nucleotides (e.g., ribavirin), immunosuppressive agents (e.g., tacrolimus), disease-modifying antirheumatic agents (e.g., leflunomide), hormone-based therapies (e.g., estradiol), and other non-neoplastic agents.3
HDs pose risks not only to patients receiving them therapeutically but also to the health care personnel who compound, administer, transport, dispose, and/or otherwise handle them.1,2 Exposure to such drugs can occur through skin and mucosal membrane absorption, inhalation, incidental ingestion, or via needle stick. HD exposure may, in turn, cause adverse effects including skin rashes, infertility, and cancer.2,4–6
Thus, preventing exposure of health care workers to HDs is of utmost importance for maintaining a safe working environment. This can be achieved through the use of engineering controls, personal protective equipment (PPE), and administrative controls.2 Closed system transfer devices (CSTDs) are one type of engineering control. These needleless devices allow HD manipulations to occur within a closed system, thus protecting health care workers from undue exposure.
If a CSTD is designed well, manufactured properly, and used appropriately, it should protect health care workers from HD exposure during the compounding and administration of HD products. All CSTD syringe adapters require the use of a syringe, which depending on its design, may compromise the closed nature of a CSTD system. The open barrel of a standard syringe can potentially lead to environmental contamination, and thus danger to health care workers. The extent of contamination possible is dependent on the drug used and its volatility, concentration, viscosity, and affinity for the syringe surface.7,8
Unlike standard open barrel syringes, sealed barrel CSTD syringe units are designed to provide a completely closed system. The typical use of any HD requires filling a syringe with the drug and transferring it to an intravenous (IV) bag or IV administration line. During the process of drawing a HD from a vial into a syringe, the HD comes into direct contact with the inner wall of the syringe for a period of time. Through this exposure, the HD may adhere to the syringe surface by chemical affinity or cohesive–adhesive forces. After the drug is transferred from the syringe, the inner surface—and any residual HD adhering to the inner surface—becomes exposed to the environment.
Potential contamination of the working environment with the HD may occur by two possible routes: evaporation of the HD or direct contact with the inner wall of the syringe. The latter type of contamination could then be spread to the working environment or health care worker via gloves or direct contact with other surfaces. This method of contamination should be prevented as much as possible and ideally, should not occur during the handling of any HD. Different levels of contamination may be observed with different HDs due to the unique physical and chemical properties of each drug; however, any level of HD contamination is a reason for concern.
Studies using CSTDs in the compounding and administration of HDs have shown a significant reduction in surface contamination levels.9,10 However, detectable levels of HDs have been observed with the use of some CSTDs. This suggests that some systems are not entirely closed or if the system is closed, there are other ways people can become exposed. Ultimately, health care workers remain at risk of exposure with their use.10,11 One study using a surface monitoring technique explored environmental contamination via syringe plunger contamination during routine drug preparation in hospital pharmacies.7
Contamination by CP on a standard open barrel syringe plunger was confirmed, localized, and quantified. Result from additional studies have confirmed the transfer of CP to a standard syringe plunger.8,9 In these studies, drug residuals on the syringe plunger contaminated both gloves and the work environment.
The purpose of this study was to quantify HD contamination of the inner surface of standard open barrel syringes and to compare contamination levels between three commonly used HDs: 5-FU, CP, and IF.
2. Methods
Three common HD products were prepared under real world compounding conditions to measure contamination levels of the inner walls of standard open barrel syringes. Using a modified NIOSH performance protocol for CSTDs,12 a total of 50 mL of drug was transferred from a vial to a 50 mL open barrel syringe, and then from the syringe to an IV bag, using the appropriate vial, syringe, and IV bag CSTD connectors. The drugs evaluated were 5-FU (50 mg/mL), CP (20 mg/mL), and IF (50 mg/mL). Becton, Dickinson and Company (BD) PhaSealTM CSTDs were used for each of the drug transfer manipulations.
The research team was comprised of a pharmacy school faculty member with extensive cleanroom experience, a pharmacy student with aseptic technique training, and a senior research associate with a doctorate in pharmacy. United States Pharmacopeia General Chapter <800> standards for protecting health care workers from HDs were adhered to throughout the testing (e.g., use of PPE, ventilated hoods, and biosafety cabinets).13
ChemoGLOTM HDClean Wipes were used for sampling the inner surfaces of the syringes, and all data were recorded on the ChemoGLOTM Site Map Form.14 Once completed, the ChemoGLOTM Site Map Forms, along with the correspondin wipe samples, were submitted to the ChemoGLOTM laboratory for analysis (Chapel Hill, North Carolina).
For each test, a CSTD vial adapter was attached to a vial containing the HD being tested, a CSTD bag adapter was attached to an IV bag, and a CSTD syringe adapter was connected to a standard open barrel syringe. The drug was then reconstituted according to the manufacturer’s instructions, if needed (i.e., CP and IF). The syringe was then attached to the vial via the CSTD adapters, and 50 mL of drug was drawn into the syringe. The 50 mL of drug was then injected into the IV bag via the CSTD IV bag adapter. Once each IV bag was prepared, the syringe barrel was tested for the presence of HD contamination.
To test the inner surface of each syringe used, a ChemoGLOTM wipe was used to wipe all four quadrants of the syringe barrel according to the following process:
- A quarter from the plunger barrel knob was removed,
allowing for controlled and easy access to the syringe
barrel without interference from the syringe plunger.
This allowed for wiping the exposed inner wall of the
syringe. - A wipe was placed into the open section, and a wooden
rod was used to move the wipe up and down. - The syringe plunger rod was rotated 90 degrees, and the
process was repeated to ensure that the entire syringe
barrel was wiped. - An additional ChemoGLOTM wipe was used to swab
each syringe quadrant a second time, using the same
method. - Each wipe was then packed and labeled according to the
instructions provided in the sampling kit.
Each test was repeated 15 times for each of the three drugs, for a total of 45 tests. The sample size of 15 syringes was based on sample sizes used in similar studies.7,8 The use of a full 50 mL per injection was also based on previous studies.7,8
Positive controls for each drug were also tested by inoculating a syringe barrel with the drug, followed by wipe sampling the inner wall of the syringe using the same method described above. No negative controls were tested since the ChemoGLOTM wipe sampling procedure is a validated process that does not require a negative sample.15
The lower limit of quantitation of the ChemoGLOTM assay is 10.0 ng/ft2 (0.011 ng/cm2) per drug, and the upper limit of quantitation (ULQ) is 4000.0 ng/ft2 (4.31 ng/cm2).16 The total drug amount found on each syringe tested was determined by adding the amounts from the two wipes used for sampling (wipe 1 plus wipe 2).
Since there were three independent groups of data and the 5-FU and IF data were not normally distributed, a Kruskal–Wallis test using a 0.05 significance level was applied to the ChemoGLOTM test results to determine if there was a statistically significant difference in contamination levels between the three HDs tested. Statistical and, descriptive analyses were performed using GraphPad Prism 10.0.2 (232) software.
Results
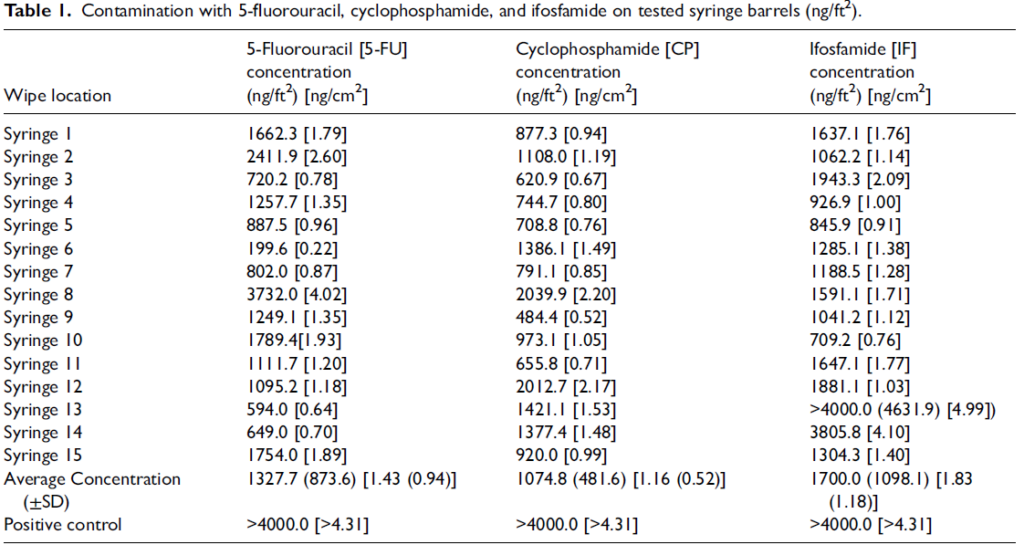
The results of this study found the inner surfaces of all 45 syringes contaminated with the HD being tested. The average amount of 5-FU detected by the ChemoGLOTM wipe kit for the 15 syringes tested was 1327.7 ng (standard deviation [SD]=873.6 ng). The average amount of CP detected was 1074.8 ng (SD =481.6 ng), and the average concentration of IF detected was 1700.0 ng (SD=1098.1 ng). The positive controls for each drug resulted in measurements exceeding 4000.0 ng each, indicating amounts beyond the test’s ULQ. Based on previous work by Cox et al., the percent recovery of the drug on each surface is estimated to be >95%.15 See Table 1 for a complete list of the data collected.
The Kruskal–Wallis test performed on the data revealed that there was no statistically significant difference between the different drugs’ level of contamination (p=0.14).
Discussion
The results from this study found significant contamination by each of the three drugs on the inner walls of the open barrel syringes. The difference in level of contamination between the three drugs was not statistically significant, highlighting that inner surface contamination by most HDs is likely when open barrel syringes are used during compounding and administration. Any differences that would exist between different HDs would likely be due to the different chemical and physical properties of the drugs —such as their polarity, hydrogen bond donor and acceptor count, and viscosity—and their relative affinity for the syringe surface. A higher SD, as is the case of IF, indicates greater variability in contamination levels.
These findings align with results from other studies. One study that assessed the extent of CP contamination on syringe plungers showed contamination in amounts ranging from 3.7 to 445.7 ng when tested via gas chromatography/ mass spectrometry (GC/MS).7 Another study, using ChemoGLOTM wipe test sampling, found CP contamination levels greater than 2000 ng on open barrel syringe plungers and no detectable contamination on sealed barrel syringe plungers after a 50 mL aliquot of CP was drawn into each syringe and injected back into the CP vial multiple times.8 The difference in this study’s CP results from these two studies’ results is likely due to the differences in analytical tools used (GC/MS vs. ChemoGLOTM wipe tests), the number of times CP was drawn into each syringe (multiple times vs. once), and/or the sampling techniques used.
One of the limitations of this study was that the positive controls for each drug resulted in measurements of greater than 4000 ng each. Since the ChemoGloTM assay has an ULQ of 4000.0 ng, the true amount of drug could not be determined for the controls. The amount of residual drug could have been anywhere from 4000 ng to orders of magnitude more. Therefore, even though the contamination of the syringe barrels from these three drugs can be quantified and compared via this testing method, the full clinical significance of the results cannot be determined by this
study alone.
Additionally, a full 50 mL of drug was drawn into each 50 mL syringe to maximize exposure of the syringe’s inner surface to the drug. This type of usage is not standard compounding practice and may have caused an overestimation of the amount of residual drug that would typically be left on the syringe inner wall during sterile product preparation or administration. Therefore, these results may not be fully generalizable to common compounding practices.
The difference in concentration of the CP solution versus the 5-FU and IF preparations could also have affected the relative amount of drug adhering to the syringe wall. However, the drug preparations used in this study (50 mg/mL for 5-FU, 20 mg/mL for CP, and 50 mg/mL for IF) are standard compounding concentrations, so these results reflect real-world comparisons, enhancing their generalizability.
Finally, only three HDs drugs were tested in this study, allowing for the possibility that additional HDs with different chemical and physical properties could produce different results.
Additional studies are warranted to analyze the extent of contamination after multiple transfers and with extended duration of use, both of which may increase the potential for HD exposure. Understanding the extent of contamination with such use would better reflect the risks associated with real-world compounding practices. Also, further studies examining the extent of transfer of HD from a syringe inner wall to a user’s gloves and compounding working space for these three drugs and other HDs are needed to better understand the extent of risk to health care workers with the use of open barrel syringes.
Conclusion
This study underscores the presence of HD contamination on standard open barrel syringe inner surfaces after transfer of drug from vial to syringe to IV bag. The detected amounts of each the three drugs (5-FU, CP, and IF) on the inner surface of standard open barrel syringes were high (ranging from 1074.8 to 1700.0 ng), especially given that the maximum amount measured (for one sample of IF) exceeded the ULQ set by the ChemoGLO assay at 4000.0 ng. Such levels of drug contamination are of concern since they could be transferred to the working environment and expose health care workers to harm. Identifying ways to limit contamination and exposure is important for the safety of all health care workers who regularly handle HDs.
Authors’ contribution
BTB and SFE conceived the study and were involved in protocol development and data collection. All authors researched literature and performed data analysis. LTA wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Declaration of conflicting interests
Equashield® provided the funding and proposed the general framework of the study. SFE and LTA have also received funding support from BD, Daiwa Can Company, and Shandong Ande Healthcare Apparatus Co., Ltd. for additional CSTD-related research. SFE is a co-founder of ChemoGLOTM. The authors declare no additional conflicts of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Equashield®.
ORCID iD
Lori T Armistead https://orcid.org/0000-0002-4680-0156


