1 Introduction
Recently, the number of marketed Closed System Transfer Device (CSTD) models has increased. Interest in development of a CSTD performance test protocol originated from within the healthcare industry itself, with requests for an independently-developed containment test protocol. Additionally, with the approval of USP chapter 800 mandating the use ofclosed systems for administration_ proper evaluation ofCSTD connectors is essential since vast majority ofadministration procedures involves exclusively the use ofCSTD connectors. To date, several leakage studies have been performed to show whether or not different brands of CSTDs are free ofleaks, drips, microbeads and drug residuals. However, most of these studies are performed on drug surrogates via a litmus paper, UV light, etc. This protocol will test CSTDs with actual antineoplastic agent Fluorouracil (5-FU}.
2 Study Objectives
Objective of this study was to test 6 different CSTD devices to assess how it matches up with their claims of being leak-proof. CSTDs were tested for 5-FU leakage detection.
Administration phase simulation:
3 Study Design
The Litmus test was performed per protocol. There were 6 brands of CSTDs evaluated in this study. 10 unique devices from each brand of CSTD were tested. 3 connector membrane or /uer activations were made per device with 5 Fluorouracil and drug was transferred back and forth between activations. Following the activations connector surfaces were tested for drug residue.
All devices were allowed to go through 1′1 membrane activation without any litmus detection. The litmus test was executed on 2nd and 3’d membrane activation.
Note that 5-FU was chosen due to its wide usage in oncology, low cost and good visibility on litmus paper. While 5- FU is in the pH range of 10, if desired the test is expandable with same materials and methodology to test additional drugs in same pH range or in the acidic pH range (preferably pH 2-4). Also, handling and cutting of litmus paper was done with nitrile gloves.
Additionally, only pharmacists or pharmacy technicians skilled in use of the tested CSTDs performed this test in accordance to protocoland manufacturers’ directions for use.
4 Supplies Needed
For assessment of Litmus Testing with 5-FU the following supplies were used:
5 Study Procedures
5.1 Negative and positive controls
For the negative control procedural steps are followed:
- A 5-FiourauracillOml vial was capped with a CSTD Vial Adapter
- A lOml syringe was attached to a mating CSTD Syringe Adapter (if needed)
- One piece of litmus paper was dipped at least half of it into sterile water for Irrigation. The wet litmus paper was padded on an absorbing pad to remove excess water droplet.
- This padded litmus paper was backed with a slight finger press on each membrane of the two mating components of the CSTD system. Sufficient distance was kept on the litmus paper between the t wo tested membranes. The purpose of rubbing with wet litmus stripe and the two twist motions is to simulate a membrane disinfection procedure with an IPA pad, a quarter turn left and quarter turn right.
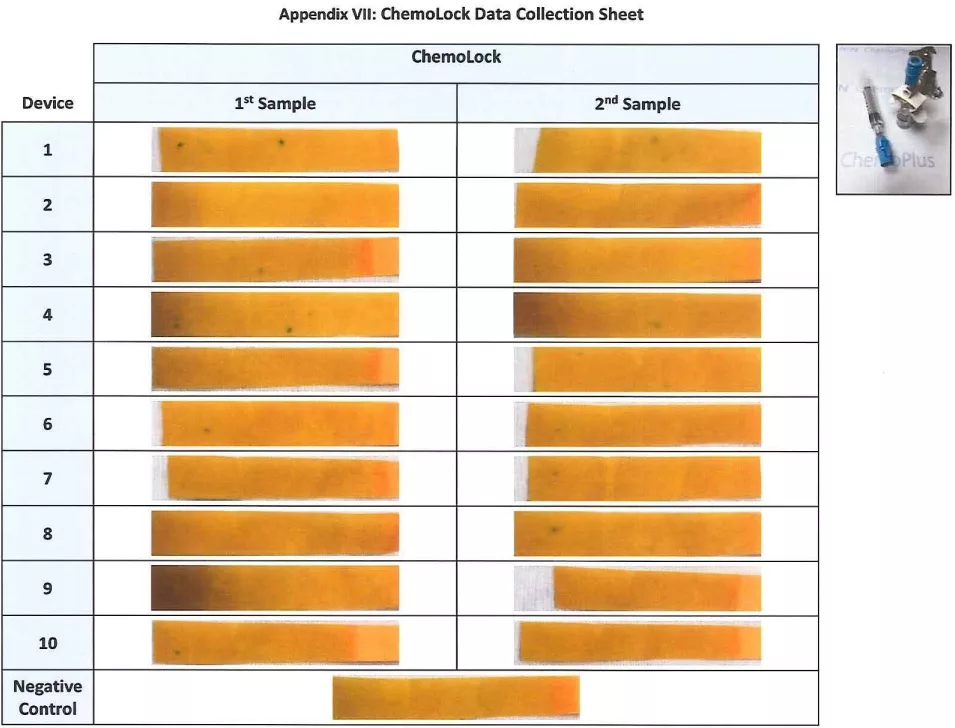
- Immediately a photograph of the negative sample was taken and denote ‘–‘ if no color change was determined and ‘y’ if color change was determined.
- Pass criteria for the negative test is if no color change was determined.
- One negative control test was performed for each brand of CSTD tested.
- If no color change was determined the negative control vial, syringe and CSTD were deemed appropriate to be used for the litmus drug test.
For the positive control procedural steps are followed:
- A 5-Fiourauracil10ml vial was accessed and a small amount of the drug was placed on the litmus paper.
- Pass criteria for the positive test is if color change was determined.
5.2 Study Procedurefor Litmus Drug Test
The following procedural steps were followed:
- A 5-Fiourauracil10ml vial was capped with a brand of CSTD Vial Adapter
- A 10ml syringe was attached to a mating CSTD Syringe Adapter (if needed)
- The syringe was attached to the vial.
- A 7ml of total volume of drug was pulled by the process of Pull-Push-Pull to simulate bubbles removal: pull 4ml, push back 4ml and pull 7ml
- The vial was inverted upright to reinject 5ml back into the vial (2mlleft in the syringe).
- The two mating systems were disconnected
- The syringe was attached to the vial and the remaining 2ml was injected from the syringe into the vial.
- Steps 4 to 6 were repeated.
- One piece of litmus paper was dipped at least half of it into sterile water for irrigat ion, t hen patted dry onto an absorbing pad to remove excess water droplet.
- The wet litmus paper was backed with a slight finger press on each membrane of the two mating components of the CSTD system. Sufficient distance on the litmus stripe was kept between the two tested membranes. The purpose of rubbing with wet litmus stripe and the two twist motions is to simulate a membrane disinfection procedure with an IPA pad, a quarter turn left and quarter turn right.
- Immediately photograph of each sample was taken and denote ‘-‘ if no color change was determined and ‘y’ if color change was determined
- Process steps 7 to 11 were repeated with the same CSTD (for a total of 3 activations)
- Test were repeated for 9 additional devices within the CSTD category with 9 additional vialsof 5-FU
- Test were completed for 5 additional CSTD brands and results recorded into data collection sheet with image capture
6 Results
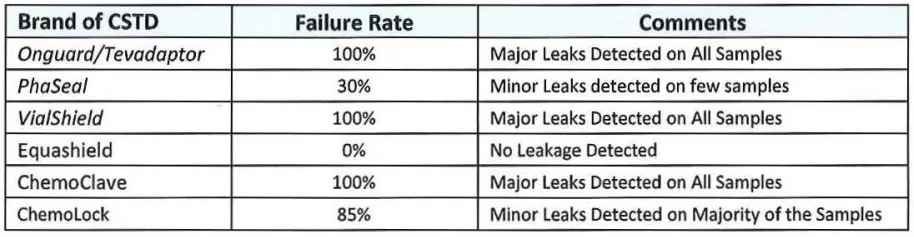
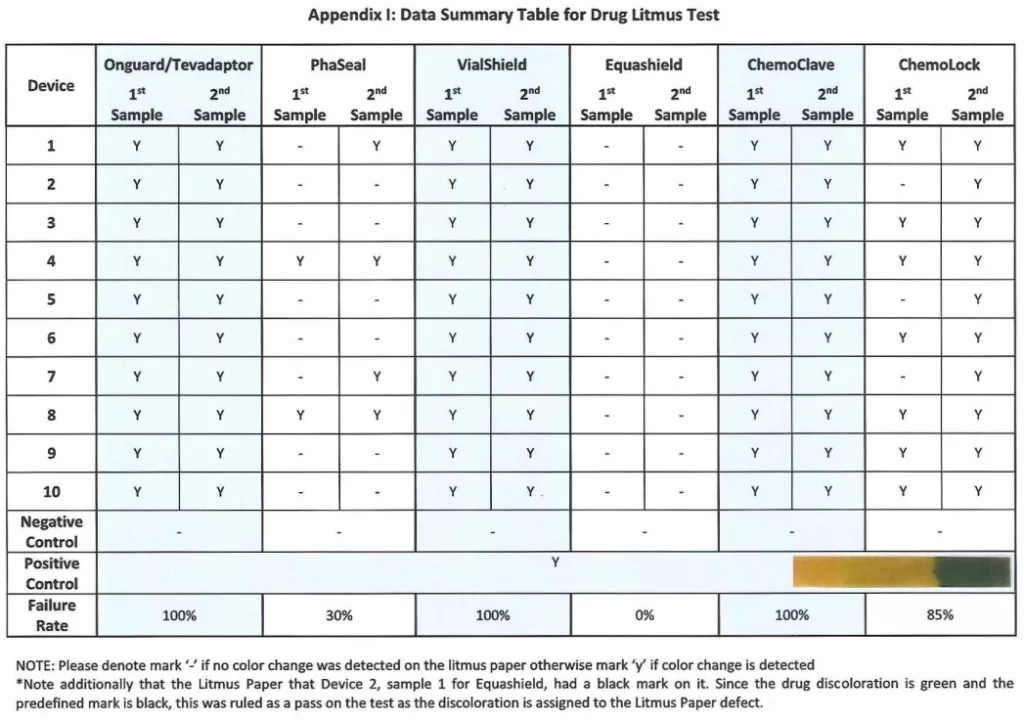
The test has been performed without any adverse occurrences. No product or procedure failures were noted. The results are clear and consistent throughout testing of the same CSTD system. The test sensitivity allows clear differentiation between performances of various CSTD systems. Of all the CSTDs brands tested, Equashield brand of CSTD was able to withstand membrane activations and showed 0 leaks. Our expectations that the test is easily replicable by any hospital pharmacy were met.
Table 1: CSTD Study Plan
Summary data is presented below:
7 Appendices
Appendix I: Data Summary Table for Drug Litmus Test
Appendix II: Onguard/Tevadaptor Data Collection

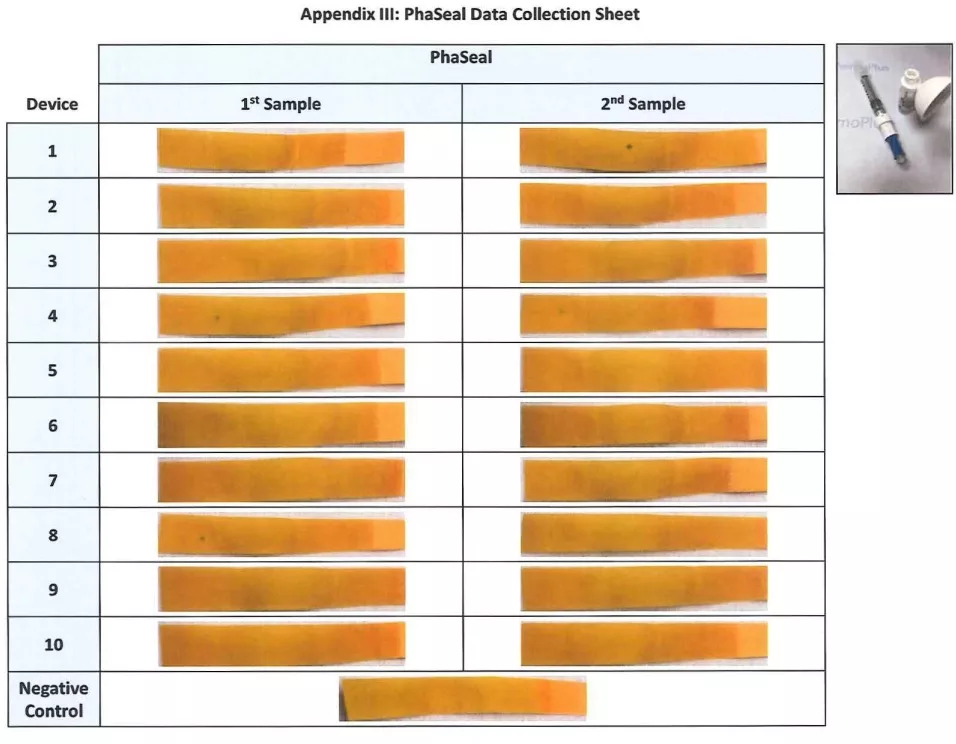
SheetAppendix Ill: PhaSeal Data Collection Sheet
Appendix IV: ViaiShield Data Collection Sheet

Appendix V: Equashield Data Collection Sheet

Appendix VI: ChemoCiave Data Collection Sheet

Appendix VII: Chemolock Data Collection Sheet